With the development of large-scale machine tools in the direction of high speed, heavy load, high precision and automation, improving the working conditions of machine tool guides, how to improve and maintain the accuracy of machine tool guides has attracted increasing attention. Because cast iron guide rails have good damping and wear resistance, stability, and low cost, they have always been the first choice of most machine tool designers. However, cast iron has weaknesses such as low hardness, loose structure, and many blank defects. Large-scale machine tools with heavier load are likely to cause wear and tear on the guide rail, which reduces the accuracy of the machine tool and affects product quality. Therefore, it is necessary to improve the wear resistance of the rail surface and the rapid repair of surface defects. Practice has proved that surface quenching and brush plating methods to protect the guide rail and brush plating-brazing method to repair the damage of the guide rail are the ideal means at present.
1. Guide rail electrical contact surface quenching
The main principle of electrical contact surface hardening is: use an electrode (graphite or copper roller) to closely contact the workpiece (Figure 1), through the low-voltage and high current of the roller, resistance heat is formed on the contact surface of the electrode and the workpiece to quickly heat the contact surface, and The surface of the workpiece is rapidly cooled by compressed air to ensure that extremely fine martensite and flake graphite are formed on the surface of the guide rail to achieve the purpose of surface quenching. At the same time, the roller moves forward at a certain speed, so that the entire guide rail can be surfaced. Quenching requirements, after actual tests, the quenching layer can reach 0.3-0.4mm and the hardness is 59-61HRC by using this method. After quenching, the surface is smoothed with a grinder, which fully guarantees the design requirements of the machine tool guide. Due to the small deformation of the guide rail, low investment, simple operation and fast speed, the economic benefits are very obvious. At present, the flat rails and V-shaped rails of the large-scale machine tools of the group company adopt this quenching and grinding method, which is simple and effective. One of the quenching machines can complete the surface quenching of 3 to 5 machine tool rails with a length of 12 to 15 m in a day.
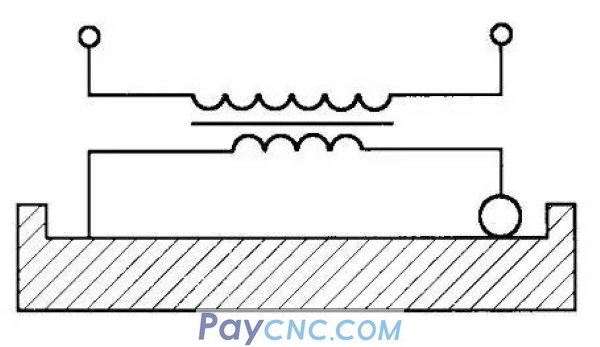
Second, use brush plating to protect the machine tool rails
Brush plating metal or alloy on the surface of the guide rail as a working layer can also strengthen its surface hardness, improve wear resistance, and further reduce the roughness requirements. Generally, quick nickel, nickel tungsten alloy, and nickel tungsten D alloy are used as the working layer. Among them, the rapid nickel alloy electrodeposition speed is fast, the hardness is equivalent to that of No. 45 steel, and the wear resistance is better than that of quenched No. 45 steel; the properties of nickel diamond tungsten alloy and nickel tungsten D alloy are similar, and both have high hardness (58~60 HRC) , The wear resistance is twice that of No. 45 steel, but the electrodeposition speed is slower than that of fast nickel. Therefore, for general medium and small machine tools, the fast nickel plating can achieve the purpose of protecting the guide rail, and the requirements for large machine tools or precision are higher. For machine tools, it is ideal to use nickel-tungsten D alloy or nickel-tungsten alloy working layer.
1. Brush plating formula
(1) Quick nickel formula. Nickel sulphate 254g/L; money salt 56g/L; money acetate 23g/L; oxalic acid 0.1g/L; ammonia (25%-28%) 105mL; specific gravity 1.15; power consumption coefficient 0.104A•h/dm2• μm.
(2) Nickel-Tungsten D alloy formula. Nickel sulfate 393g/L; sodium tungstate 23g/L; boric acid 31g/L; citric acid 42g/L; sodium sulfate 6.5g/L; cobalt sulfate 2g/L; additives 2.8g/L; glacial acetic acid 20ML/L; Sodium fluoride 5g/L; sodium lauryl sulfate 0.01~0.001 g/L; pH value 1.4~2.4; power consumption coefficient 0.214 A•h/dm2•μm.
(3) Electric brush plating pretreatment process. Industrial gasoline cleaning the surface of the guide rail → water sandpaper polishing → acetone fine washing → electric degreasing (voltage 12V, machine tool connected to the negative electrode) → water washing → No. Ⅱ electric cleaning (workpiece connected to the positive electrode, voltage 7V) → water washing → No. Ⅲ electric cleaning (workpiece connected Positive electrode, voltage 10~14V, and then connect the workpiece to negative electrode, voltage 6~8V)→wash with water. Special nickel is used as the transition layer of electric brush plating, water washing, electric brush plating of quick nickel or nickel tungsten D alloy or nickel drill tungsten alloy.
2. Brush plating anode material and plating solution circulation
(1) Anode material. At present, graphite is generally used as the anode in electric brush plating. Graphite has good conductivity and no other metal ions pollute the electroplating solution. However, due to the brittleness of graphite, it is prone to collapse under high-density current. At the same time, the strength of graphite is low and it is not easy to make large electrodes. Metal is easy to form and has high strength, so high-purity metal can be used as anode, but metal as anode is likely to cause a large amount of metal ions to be incorporated into the electroplating solution. Therefore, the electroplating solution must contain a complexing agent with strong complexing force to complex excess metal ions. To ensure the stability of the electroplating solution and to prevent the passivation of the metal anode under high-density current. Fast nickel brush plating, nickel tungsten D alloy or nickel diamond tungsten alloy all use electrolytic metal nickel as the anode.
The anode made by should be flat, the conductive plate and the nickel plate should be uniformly spot welded with stainless steel electrodes, and a 3mm plum-shaped small hole should be drilled on the nickel plate at the bottom of the anode. The electric brush plating anode is shown in Figure 2.
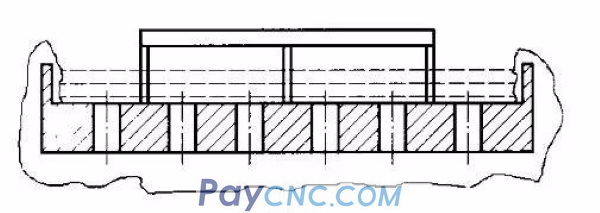
(2) Plating bath circulation. The closed method and the intermittent pouring method can be used. Sealing method: the plating solution is enclosed in the tank, and the anode is immersed in the plating solution for brush plating. At this time, the brush plating layer is soaked in the plating solution. The plating solution is not easily oxidized by air and affects the quality of the brush plating. It is generally suitable for An alloy electroplating solution that is more expensive and has a slower electrodeposition rate. Intermittent pouring method: The electroplating solution is poured on the anode intermittently to make it naturally consumed. In order to reduce the loss of the electroplating solution, a groove can be processed on the back of the anode, and a small hole is drilled at the bottom of the anode, and the plating solution penetrates from the small hole. Coating on absorbent cotton, this method has a lot of electroplating liquid loss, and is generally used for electric brush plating with high speed and relatively cheap electroplating liquid.
(3) Coating thickness control. The formula for calculating the thickness of the coating: δ=I•t/C•S. Where δ is the thickness of the coating (μm), I is the current (A), t is the time (h), S is the area (dm), and C is the power consumption coefficient of the plating solution (A•h/dm2•μm). The more uniform the thickness of the coating, the higher the accuracy of the guide rail. The uniformity of the coating is affected by the shape of the anode, the uniformity of the anode sheath thickness, and the conductivity of the anode. Generally, the thickness of the electroplating layer is 0.05 to 0.08 mm, and the unevenness can be controlled within 0.003 mm. Inside.
3. Repair the guide rail damage by brush plating and brazing method
Cast iron has high carbon content, loose structure, and poor bonding force directly on the brazing. For this reason, nickel can be used as the transition layer and alkaline copper as the intermediate layer, in which nickel is used as the transition layer or the bottom layer has good adhesion to the matrix. As the intermediate layer, it can be firmly combined with nickel, and can be combined with the brazing layer of tin alloy. The tin alloy has low melting point, good fluidity, toughness, and wear resistance. At the same time, its hardness and melting point can be changed according to the alloy composition. Adjust within a certain range and combine the actual situation to formulate the specific process as follows:
1. Surface treatment
The surface treatment of the guide rail (removal of oil and dust). Electric degreasing → water washing → Ⅱ electric cleaning → Ⅲ electric cleaning → brushing transition layer nickel 2~3μm → water washing → brushing alkali copper plating → water washing → pickling → brazing tin secret alloy → smoothing the brazing layer → brushing pretreatment →Brush plating.
It is especially emphasized that the oil, ash, and sand on the damaged part of the guide rail must be washed with gasoline and the serious fatigue layer must be removed before brushing, otherwise it will seriously affect the bonding strength of the welding layer. It is easy to oxidize when brushing alkali copper layer, and the oxide layer will affect the bonding strength of the solder layer. Therefore, the oxide film should be washed off with hydrochloric acid before welding. At the same time, the hydrochloric acid should not be applied too much, otherwise it will corrode the substrate. There are many pinholes in the solder layer of tin alloy.
2. Nickel formula and process
Nickel sulfate 330g/L; glacial acetic acid 30mL/L; glycine 20 mL/L, power consumption coefficient 0.42 A•h/dm2•μm; pH value 0.8-1; relative movement speed of cathode and anode 10~25m/min; voltage 8 ~10V.
3. Alkaline copper formula and process
Copper sulfate 250g/L; ethylenediamine 250g/L; nitrilotriacetic acid 150 g/L; sodium sulfate 20 g/L; nitric acid reamer 50g/L; pH value 7-8; relative movement speed of anode and cathode 10-20m/min ; Voltage 6~15V; Power consumption coefficient 0.18 A•h/dm2•μm.
The above methods can be used according to different conditions of machine tool manufacturing or maintenance. For newly processed machine tool rails, surface quenching or brush plating can be used; for damaged rails, brush plating and brazing can be used to repair first, and then the entire rail surface can be quenched or brushed with a layer of metal or alloy . This not only solves the repair problem, but also protects the entire machine tool rail and prolongs the service life of the machine tool rail. Atlas Air Compressor Parts Valve
|
 |
| Products Catalogue | Home | About Us | Retrofit | Download | News | Tech Support | Contact Us | |
|
|
|
